Active recall of masks and protective clothing products in recent two weeks
Health Times
The Health Times found that in the past two weeks, enterprises in Beijing, Guangdong, Henan and Anhui actively recalled masks and protective clothing products due to the unqualified mask belt and ventilation resistance, and the unqualified anti-static indicators and needle distance indicators of protective clothing.
Beijing: An enterprise voluntarily recalled protective clothing for sales nineteen thousand nine hundred and eighty-six piece
On July 19, according to the official website of Beijing Municipal Drug Administration, Beijing Penglai Pharmaceutical Co., Ltd. voluntarily recalled the disposable protective clothing for medical use. Since the appearance of the protective clothing with the batch number of 211201 did not meet the requirements after inspection by Beijing Medical Device Inspection and Research Institute, the batch was actively recalled, and the recall level was Level III. The reason for recall involves that the needle distance does not meet the requirements, but because there is a heat seal at the splice connection, it will not cause leakage. The sales volume of products involved is 19986.
Anhui: two enterprises voluntarily recalled protective clothing and medical masks for sale six hundred and thirty Pieces and twenty-four 10000
On July 8, according to the official website of Anhui Drug Administration, Anhui Tiankang Medical Technology Co., Ltd. voluntarily recalled disposable protective clothing. The disposable medical protective clothing with batch numbers of 220302 and 220303 is unqualified through sampling inspection, and the recall level is Grade III. The reason for recall involves that the anti-static index or needle distance index is unqualified, and the sales quantity of the two batches is 330 pieces and 300 pieces respectively.
In addition, on July 11, according to the official website of Anhui Provincial Drug Administration, Bengbu Huohe Pharmaceutical Co., Ltd. actively recalled the disposable medical masks. The batch number 2021061201 of the disposable medical masks failed to pass the sampling inspection, and the recall level was Level 3. This batch number sold 240000 pieces in China.
Henan: An enterprise voluntarily recalled medical masks for sale seventy-nine thousand six hundred and forty slice
On July 15, according to the official website of Henan Drug Administration, Nanyang Tianhua Pharmaceutical Co., Ltd. actively recalled the disposable medical masks. The products with the batch number of 20210902 were recalled to the third level due to the unqualified mask belt and ventilation resistance, and the domestic sales volume was 79640 pieces. In addition, the report mentioned that the company attached great importance to the recall of disposable medical masks according to procedures. During the epidemic, customers had used up the masks, and the number of recalls was zero, which did not affect personnel.
Guangdong: Two enterprises voluntarily recalled medical surgical masks for sales sixty-five thousand Only and six thousand and five hundred individual
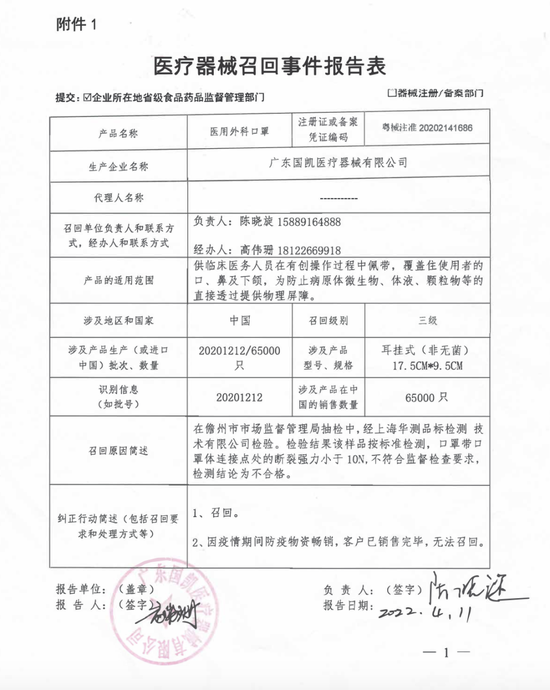
On July 18, according to the website of Guangdong Drug Administration, Guangdong Guokai Medical Instruments Co., Ltd. voluntarily recalled medical surgical masks. The surgical masks with the production batch number of 20201212 were found not in conformity with the standard after sampling inspection. The recall level was Level 3, and the sales volume in China was 65000. The reason for recall mentioned that the breaking strength of the mask belt at the connection point of the mask body is less than 10N, which does not meet the supervision requirements.
In addition, on July 18, according to the website of Guangdong Provincial Drug Administration, Guangzhou Baiyun Lantian Electronic Technology Co., Ltd. actively recalled medical surgical masks. The production batch number of 26821051 medical surgical masks was found to be not in conformity with the standard after sampling inspection. The recall level was Level 3, and the sales volume in China was 6500. The reason for recall mentioned that the breaking strength of the mask belt at the connection point of the mask body is less than 10N, which does not meet the supervision requirements.
Massive information, accurate interpretation, all in Sina Finance APP
Editor in charge: Li Siyang